Introduction
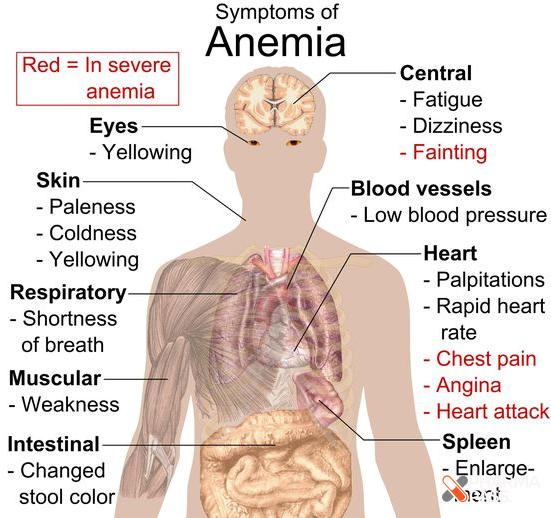
Anemia chapter is designed for student preparing for PEBC (Pharmacy Examining Board of Canada) exams. It provides a detailed overview of anemia, a condition characterized by an insufficient number of red blood cells or reduced oxygen-carrying capacity to meet the body’s physiological needs. Anemia is a widespread global public health concern, affecting an estimated 1.62 billion people worldwide. Anemia can be defined in various ways, but the World Health Organization (WHO) recommends hemoglobin (Hb) as a reliable and objective marker. According to the WHO, anemia is diagnosed when Hb levels are less than 130 g/L in men and less than 120 g/L in nonpregnant women. Additionally, reductions in hematocrit (Hct) or red blood cell (RBC) count can also indicate anemia. However, it is essential to recognize that these thresholds may not apply universally across all patient populations. For example, individuals living at high altitudes typically exhibit higher Hb levels than those at sea level due to environmental adaptations. Similarly, smokers and individuals exposed to carbon monoxide may have elevated Hct values compared to nonsmokers. Moreover, African-American populations often have Hb values that are 5–10 g/L lower than those of comparable Caucasian populations. This difference may be attributed to social determinants of health and should be interpreted cautiously to avoid misdiagnosis. This chapter focuses on anemias that respond to pharmacologic therapy, specifically those seen in the adult population. Topics covered include anemia caused by iron, vitamin B12, and/or folate deficiencies, as well as anemia responsive to erythropoietin therapy. Additionally, the chapter briefly discusses anemia of chronic disease, particularly in the context of its differential diagnosis from iron deficiency anemia.
Goals of Therapy
The primary therapeutic objectives when managing anemia are as follows:
Alleviate the Signs and Symptoms of Anemia: Reduce fatigue, pallor, and other clinical manifestations.
Restore Normal or Adequate Hemoglobin Levels: Achieve levels appropriate for the patient’s age, sex, and physiological status.
Improve Quality of Life: Address physical and cognitive impairments related to anemia.
Prolong Survival: Reduce the risk of complications associated with chronic anemia, such as cardiovascular strain.
By achieving these goals, practitioners can ensure optimal patient outcomes and improve overall health and well-being. This chapter will provide the foundational knowledge necessary for understanding anemia and its management, essential for both clinical practice and PEBC exam success.
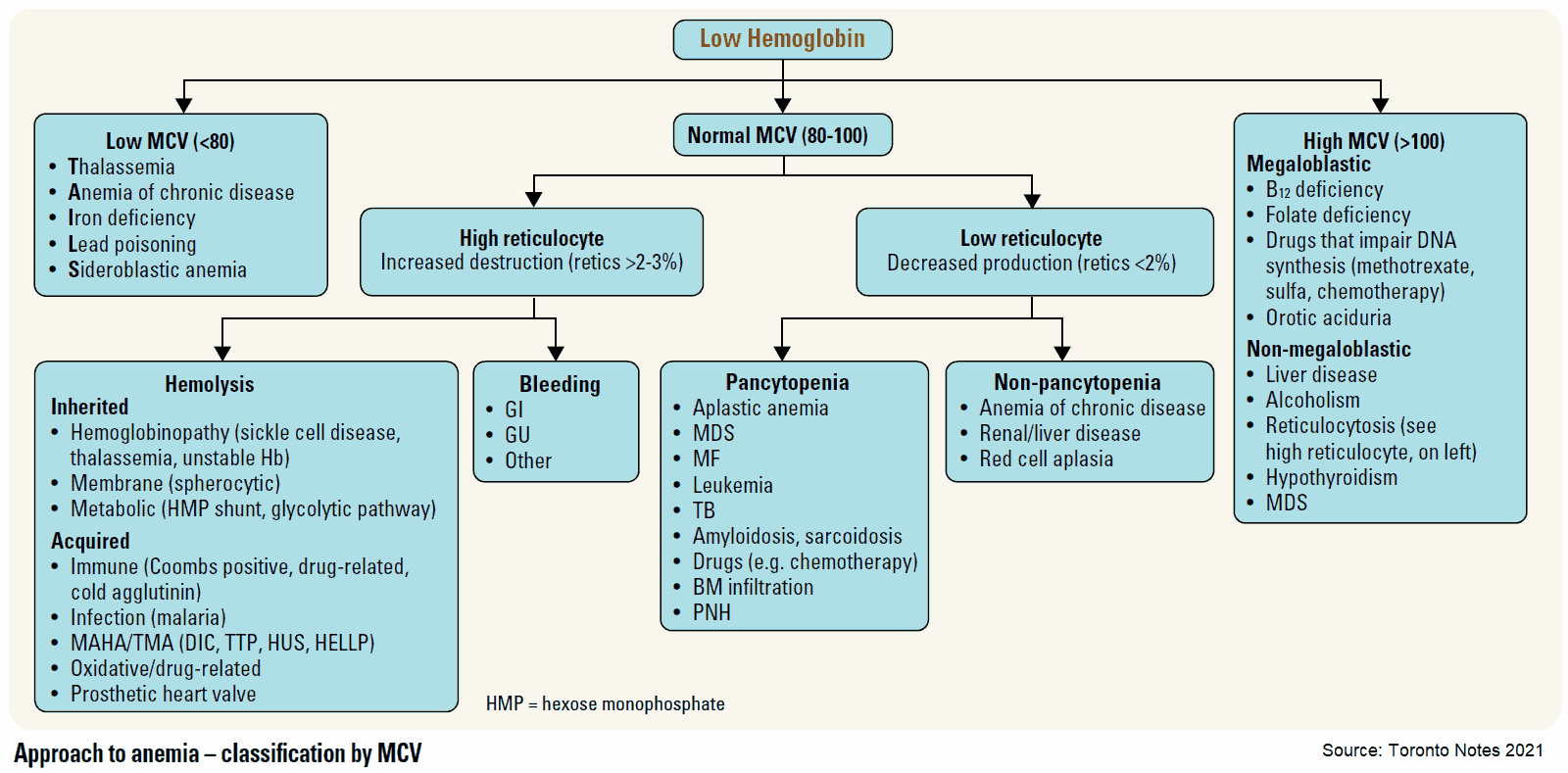
Assessment
Iron Deficiency Anemia
Iron Deficiency Anemia (IDA) is the most common nutritional disorder worldwide, accounting for nearly half of all anemia cases. It predominantly affects preschool children and women due to higher iron requirements and potential for increased iron loss. In adults, IDA is present in approximately 1–2% of the population. Additionally, iron deficiency without anemia occurs in up to 11% of women and 4% of men.
The primary cause of iron deficiency is blood loss, which can be overt (visible) or occult (hidden). In men and postmenopausal women, occult gastrointestinal (GI) bleeding is the most frequent source of iron loss. Other at-risk groups include:
Regular Blood Donors: Frequent blood donation can deplete iron stores.
Patients with Chronic Hemolysis: Conditions causing ongoing destruction of red blood cells increase iron demands.
Menstruating Women: Monthly blood loss can lead to iron deficiency, especially with heavy menstrual bleeding.
Decreased iron absorption is another factor contributing to IDA. This can occur in individuals with:
Malabsorptive Disorders: Such as celiac disease or autoimmune atrophic gastritis.
Gastric Bypass Surgery: Surgical alterations of the GI tract can impair iron absorption.
IDA typically presents as a microcytic anemia, characterized by a low Mean Corpuscular Volume (MCV). For diagnosis:
Serum Ferritin Level: A key diagnostic test; decreased levels confirm iron deficiency.
Transferrin Saturation: Can also aid in assessing iron status.
Iron metabolism is regulated by hepcidin, a hormone primarily produced by the liver. Key points about hepcidin include:
High Hepcidin Levels: Promote iron storage in the liver, spleen, and bone marrow, and decrease iron absorption from the gut.
Suppression of Hepcidin Production: Occurs in response to hypoxia (low oxygen levels), iron deficiency, and the presence of erythropoietin (a hormone stimulating red blood cell production).
Correlation with Ferritin: Increased hepcidin levels are associated with elevated serum ferritin, indicating abundant iron stores.
In IDA patients, typical findings include:
Low Serum Ferritin: Reflecting depleted iron stores.
Microcytic Red Blood Cells: Smaller than normal RBCs due to impaired hemoglobin synthesis.
Low Hemoglobin (Hb): Resulting from insufficient iron for hemoglobin production.
Low Hepcidin Levels: Theoretically expected, although not commonly measured in clinical practice.
Anemia of Chronic Disease
Anemia of Chronic Disease (ACD), also known as anemia of inflammation, is the second most prevalent cause of anemia after IDA. It occurs in the context of chronic infections, inflammatory disorders, or malignancies.
During inflammation or infection:
Hepcidin Levels Increase: Inflammatory cytokines like interleukin-6 and bacterial lipopolysaccharides stimulate hepcidin production.
Hepcidin's Role: Acts as an antimicrobial agent by reducing iron availability to pathogens, effectively sequestering iron within storage sites and decreasing its circulation.
Diagnostic features of ACD include:
Elevated Serum Ferritin: Iron stores appear normal or increased due to sequestration.
Evidence of Inflammation: Elevated serum C-reactive protein (CRP) levels.
Anemia Presentation: Typically normocytic (normal MCV) or microcytic anemia.
Laboratory tests used to assess iron disorders in ACD are:
Serum Transferrin: Often decreased or normal, reflecting reduced iron transport capacity.
Transferrin Saturation: Usually decreased due to limited iron availability despite normal or increased iron stores.
Challenges in ACD management:
Overlap with Iron Deficiency: Patients may simultaneously have iron deficiency and ACD, complicating diagnosis and treatment.
Underlying Disorder: Since ACD is secondary to another condition, addressing the primary cause (e.g., controlling inflammation) is essential but may not always be feasible.
Comparative Table of Laboratory Findings in Iron Deficiency Anemia and Anemia of Chronic Disease
To better understand the differences between IDA and ACD, the following table summarizes key laboratory findings and characteristics associated with each condition:
Nonpharmacologic Choices
Dietary changes can play an important role in managing iron deficiency anemia (IDA), but they are often insufficient to replenish iron stores rapidly, especially in cases of severe deficiency. Key aspects include:
- Heme Iron Sources: Foods rich in heme iron, such as liver, lean red meats, seafood (e.g., oysters, clams, tuna, salmon, sardines, shrimp), are better absorbed and contribute significantly to iron replenishment.
- Non-Heme Iron Sources: Found in plant-based diets, these have lower bioavailability and require more time to correct iron deficiency. Combining these foods with vitamin C–rich foods (e.g., citrus fruits, bell peppers) may enhance absorption, particularly for vegetarians or vegans.
- Factors Affecting Absorption: Polyphenols and phytates in tea and coffee can inhibit non-heme iron absorption, so these beverages should be consumed separately from iron-rich meals.
- Role of Vitamin C: Although controversial, combining vitamin C with dietary non-heme iron sources may still be a reasonable recommendation.
Pharmacologic Choices
Oral and parenteral iron therapies are commonly used to treat IDA, with oral therapy being the primary approach in most cases.
Oral Iron Therapy:
- Iron Salts: The mainstay of IDA treatment, available in various forms with differing amounts of elemental iron. Examples include ferrous sulfate, ferrous gluconate, and ferrous fumarate.
- Dosing Recommendations:
- Treatment Doses: Typically given as daily or alternate-day dosing. Alternate-day dosing has been shown to enhance absorption and reduce gastrointestinal (GI) side effects.
- Prevention Doses: Smaller doses may suffice in preventing recurrence of iron deficiency.
- Administration Tips:
- Taking iron on an empty stomach increases absorption but may lead to nausea or epigastric discomfort.
- GI side effects can be minimized by starting with smaller doses or taking iron with meals.
- Enteric-coated iron tablets are poorly absorbed and should be avoided.
Alternative Oral Options:
- Polysaccharide-Iron Complex: Provides 150 mg elemental iron per capsule and is better tolerated than some iron salts, but clinical results are inconsistent.
- Heme Iron Polypeptides: Contain 11 mg elemental iron per tablet, are derived from bovine hemoglobin, and are considered easier to absorb and tolerate. However, evidence for their efficacy in treating IDA is limited.
Parenteral Iron Therapy:
- Reserved for patients with malabsorption, intolerance to oral iron, severe anemia, or when rapid replenishment is needed.
- Characteristics:
- Consists of iron complexed with a carbohydrate or salt.
- High-dose preparations (e.g., iron isomaltoside) can replenish iron stores in 1–2 infusions.
- Requires administration in a hospital or outpatient clinic.
- Advantages: Faster rise in hemoglobin (Hb) levels compared to oral therapy, with reduced need for red blood cell (RBC) transfusions.
- Risks: Potentially increased risk of infection and rare adverse events like anaphylaxis.
Therapy During Pregnancy and Breastfeeding
Iron requirements increase during pregnancy due to the expansion of maternal red cell mass and the growth of the fetus and placenta. Treatment and prevention strategies include:
Dietary and Oral Iron:
- Dietary supplementation and oral iron salts are generally safe and effective during pregnancy.
- A dose of 20 mg/day elemental iron from 20 weeks gestation has been shown to prevent IDA and improve pregnancy outcomes.
- For patients with GI side effects, lower doses or intermittent regimens (e.g., once or twice weekly) are better tolerated.
Parenteral Iron:
- Used in cases of severe anemia, malabsorption, or oral iron intolerance.
- Systematic reviews suggest parenteral iron results in higher Hb concentrations at six weeks postpartum compared to oral iron.
- Precautions should be taken, and product-specific warnings reviewed before use in pregnancy.
Breastfeeding:
- Iron passes into breast milk, providing an essential dietary source for infants. Maternal iron supplementation does not significantly alter milk iron content.
Therapeutic Notes
Investigating the Cause: The underlying cause of anemia should always be identified and addressed. For example:
- In men and postmenopausal women, occult GI bleeding should be ruled out, as IDA may indicate a GI malignancy.
- Screen for malabsorption conditions like celiac disease if no obvious blood loss is identified.
Treatment Adjustments:
- If Hb levels do not improve as expected, consider ongoing blood loss, impaired iron absorption, nonadherence, or alternative causes of anemia.
- A graduated approach to iron dosing may help patients tolerate therapy better, particularly in those prone to GI upset.
Special Populations:
- Pregnant patients with physiologic decreases in Hb should still be evaluated for iron deficiency.
- Elderly patients and those with significant GI symptoms may benefit from low daily doses (e.g., 15–50 mg elemental iron).
Duration of Therapy:
- Continue iron supplementation until Hb levels normalize and iron stores are replenished. This typically requires 3 months of therapy after reaching the target Hb level.
- In chronic or unresolved causes of IDA, educate patients about anemia symptoms and regularly monitor for recurrence.
Table: Pharmacologic Options for Treating Iron Deficiency Anemia
| Option | Elemental Iron | Recommended Dosing | Advantages | Limitations |
|---|---|---|---|---|
| Ferrous Sulfate | 65 mg/tablet | 1–2 tablets daily or alternate-day dosing | Effective, affordable | Common GI side effects (nausea, constipation) |
| Ferrous Gluconate | 35 mg/tablet | 1–3 tablets daily | Better GI tolerance than ferrous sulfate | Lower elemental iron content |
| Ferrous Fumarate | 106 mg/tablet | 1 tablet daily or every other day | High elemental iron content | Increased risk of GI upset |
| Polysaccharide-Iron Complex | 150 mg/capsule | 1 capsule daily | May be better tolerated | Variable clinical efficacy |
| Heme Iron Polypeptide | 11 mg/tablet | 1 tablet 3 times daily (preventative dose) | Easier absorption, fewer GI side effects | Limited evidence for treating IDA |
| Parenteral Iron | Variable (high dose) | 1–2 infusions for full replacement dose | Rapid replenishment, avoids GI side effects | Requires clinical setting, risk of infection |
Anemias Responsive to Pharmacologic Stimulation of Erythropoiesis
While most patients with nutritional anemias (e.g., iron, vitamin B12, or folate deficiency), hemolysis, or bleeding naturally have elevated levels of endogenous erythropoietin, certain conditions benefit from pharmacologic stimulation of red blood cell (RBC) production using erythropoiesis-stimulating agents (ESAs). These include:
- Chronic Kidney Disease (CKD): Anemia resulting from reduced erythropoietin production.
- Chemotherapy-Induced Anemia: In patients with nonhematologic cancers.
- Preoperative Anemia: To minimize transfusion requirements in surgical patients.
- Low-Risk Myelodysplastic Syndrome (MDS): To address symptomatic anemia.
- Antiretroviral-Induced Anemia: In patients with HIV infection.
- Hepatitis C–Associated Anemia: Due to ribavirin therapy.
Assessment
Eligibility Criteria:
- Patients generally qualify for ESA therapy if hemoglobin (Hb) levels are <100 g/L.
- Baseline endogenous erythropoietin levels should also be assessed. Normal levels in healthy individuals are approximately 3–30 units/L.
Iron Supplementation:
- Adequate iron supply is essential to support erythropoiesis when using ESAs. Iron deficiency should be corrected prior to or concurrently with ESA therapy to maximize efficacy.
Pharmacologic Choices
Two ESAs are available in Canada, each with distinct pharmacokinetic properties:
Epoetin Alfa:
- Type: Recombinant human erythropoietin.
- Dosing and Administration:
- Given via IV or SC injection.
- Short half-life necessitates administration 3 times weekly.
- In preoperative surgical settings, daily dosing may be used for a more rapid rise in Hb.
Darbepoetin Alfa:
- Type: Synthetic erythropoietin analogue.
- Dosing and Administration:
- Longer half-life allows for weekly, biweekly, or monthly dosing, depending on the patient’s clinical needs.
Efficacy and Risks
Benefits:
- ESAs improve quality of life in patients with anemia due to chronic kidney disease.
- ESAs are not as effective in improving quality of life or fatigue in patients with AIDS- or cancer-related anemia.
Risks:
- Increased risk of cardiovascular events (e.g., hypertension, thrombosis) when dosed to achieve normal Hb levels.
- Pure Red Cell Aplasia (PRCA):
- Rare complication caused by the formation of neutralizing antibodies that cross-react with endogenous erythropoietin, leading to severe anemia.
- Modern formulations and handling practices have greatly reduced the incidence of PRCA, but it should still be suspected in therapy-refractory patients.
Dosing Guidelines:
- Target Hb levels should remain between 100–120 g/L, depending on the indication.
- Monitor Hb monthly to prevent excessive correction.
Choices During Pregnancy and Breastfeeding
Pregnancy:
- No well-controlled studies exist on ESA use in pregnancy.
- Risks: Potential for hypertension and thrombosis.
- Benefits: Reduces risks associated with anemia and frequent blood transfusions during pregnancy.
- Use ESAs only when the potential benefits outweigh the risks to the fetus, with adequate iron supplementation to support erythropoiesis.
Breastfeeding:
- There is no evidence of ESAs passing into breast milk. However, caution is advised when using ESAs in breastfeeding patients.
Therapeutic Notes
Judicious Use: ESAs should be reserved for patients requiring transfusion support to minimize associated risks.
Titrated Dosing: Doses should be adjusted to ensure gradual improvement in anemia without overshooting target Hb levels.
Monitoring:
- Blood Pressure:
- Rapid or excessive correction of anemia may provoke hypertension or seizures, particularly in susceptible individuals.
- Monitor blood pressure three times per week initially and after each ESA dose thereafter.
- Thrombotic Complications:
- Erythrocytosis (excessive RBC production) can predispose patients to thrombosis.
- Blood Pressure:
Patient Education: Inform patients about the risks of ESAs, the importance of iron supplementation, and adherence to prescribed dosing schedules.
Table: Comparison of Erythropoiesis-Stimulating Agents
| Agent | Half-Life | Dosing Frequency | Advantages | Limitations |
|---|---|---|---|---|
| Epoetin Alfa | Short | 3 times weekly (or daily pre-op) | Effective for rapid Hb improvement | Requires frequent dosing |
| Darbepoetin Alfa | Long | Weekly, biweekly, or monthly | Longer intervals between doses | Higher cost, slower Hb response |